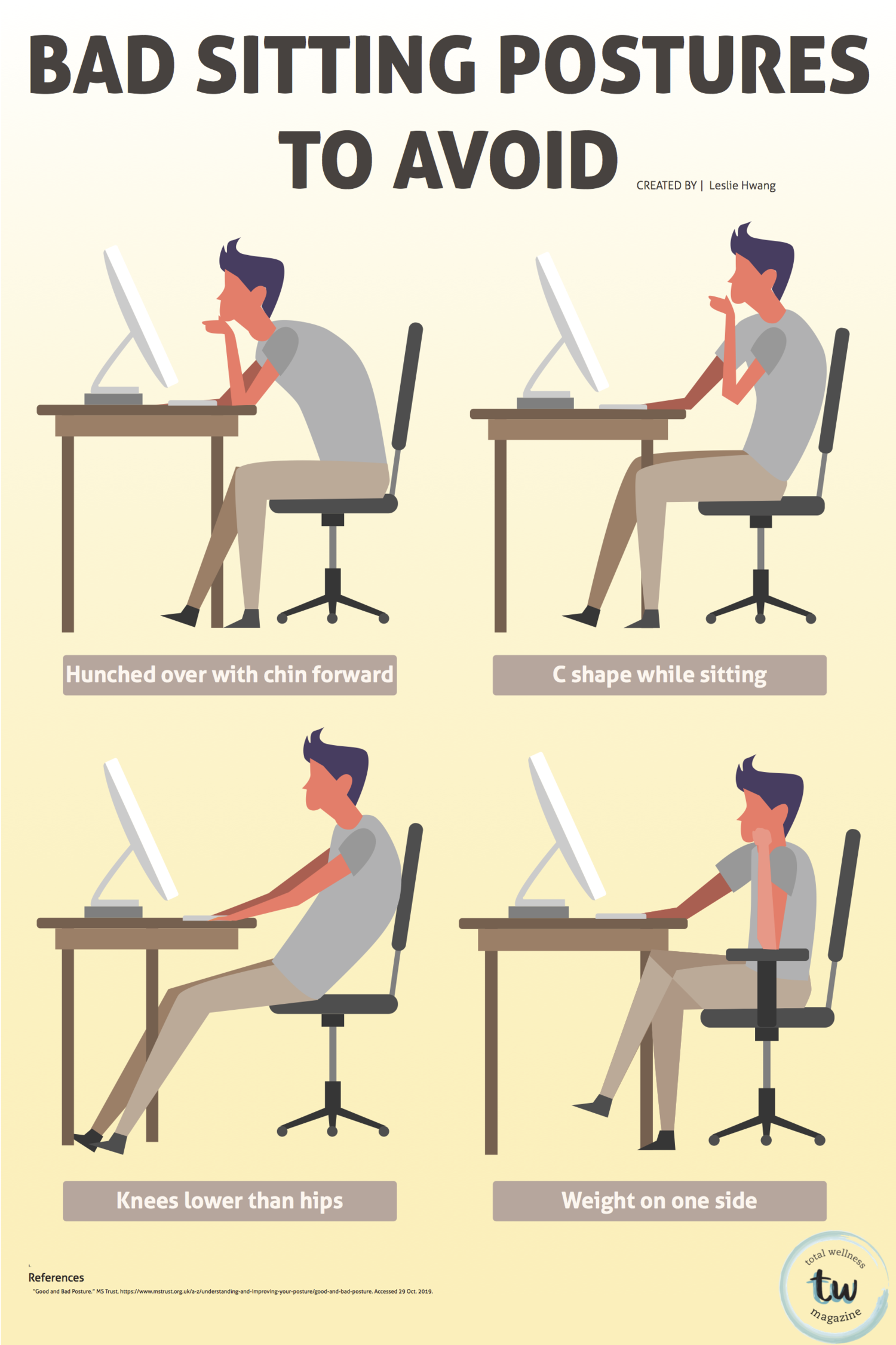
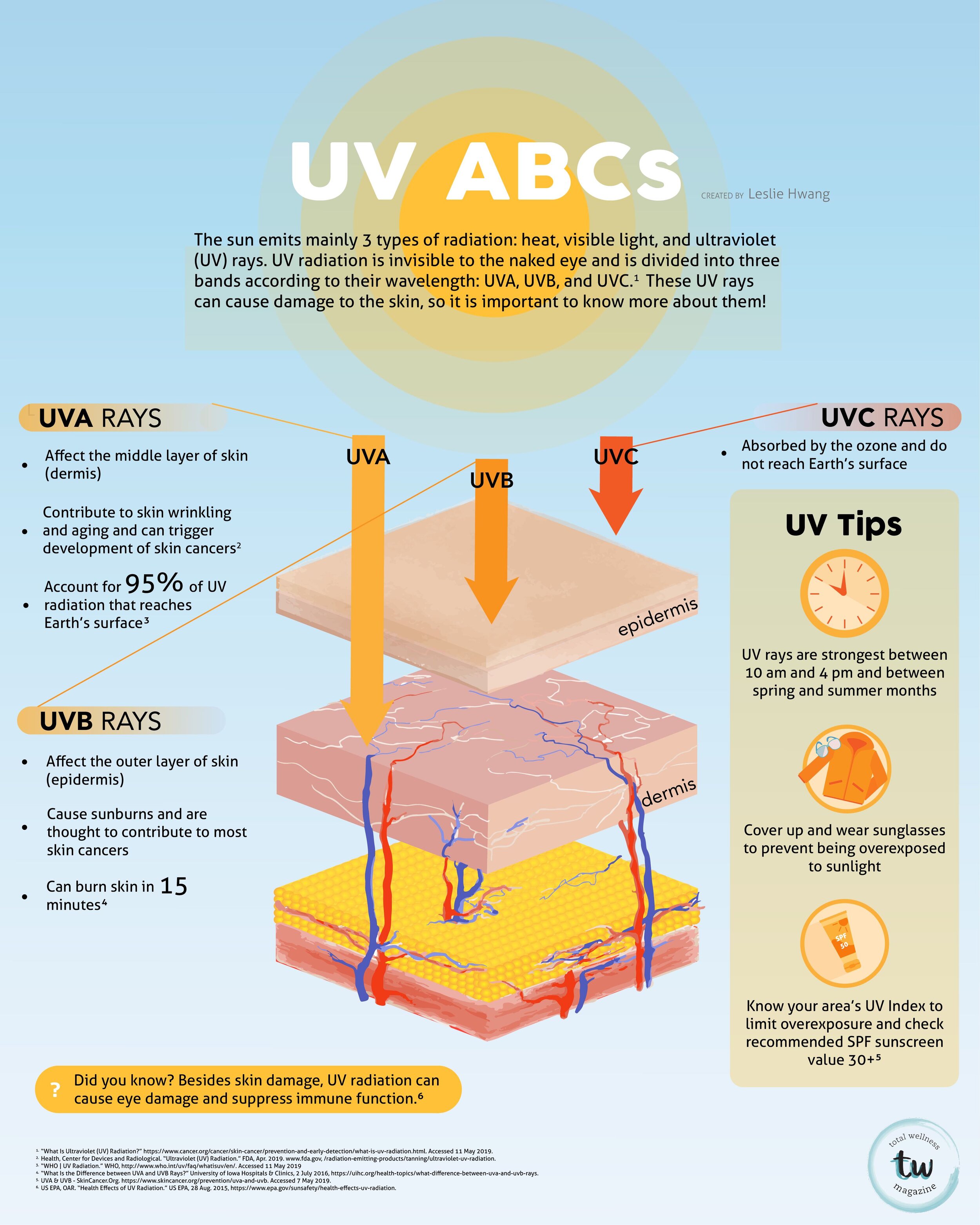
FEATURED ARTICLES
Articles

Eat Well
5 Foods That Boost Your Immune System
Eat Well
Eat Well
Latest articles
Articles
bruin EXCLUSIVE
BRUIN DESIGNS
![Lunar New Year [UCLA Total Wellness].png](https://images.squarespace-cdn.com/content/v1/56a19dc589a60afe613cef70/1579988671858-BWEP8ZRA6I7FQRFGUKKC/Lunar+New+Year+%5BUCLA+Total+Wellness%5D.png)
![Protein Foods [UCLA Total Wellness].jpg](https://images.squarespace-cdn.com/content/v1/56a19dc589a60afe613cef70/1579989281745-LDS4SSNBKKQKBN9IS7YT/Protein+Foods+%5BUCLA+Total+Wellness%5D.jpg)







![Choco-Banana-Smoothie-[Rachel-Tsao].png](https://images.squarespace-cdn.com/content/v1/56a19dc589a60afe613cef70/1549377428668-0IL5RLNN7H4WV389BMC9/Choco-Banana-Smoothie-%5BRachel-Tsao%5D.png)

![Lunar New Year [UCLA Total Wellness].png](https://images.squarespace-cdn.com/content/v1/56a19dc589a60afe613cef70/1579988671858-BWEP8ZRA6I7FQRFGUKKC/Lunar+New+Year+%5BUCLA+Total+Wellness%5D.png)
![Protein Foods [UCLA Total Wellness].jpg](https://images.squarespace-cdn.com/content/v1/56a19dc589a60afe613cef70/1579989281745-LDS4SSNBKKQKBN9IS7YT/Protein+Foods+%5BUCLA+Total+Wellness%5D.jpg)









![Choco-Banana-Smoothie-[Rachel-Tsao].png](https://images.squarespace-cdn.com/content/v1/56a19dc589a60afe613cef70/1549377428668-0IL5RLNN7H4WV389BMC9/Choco-Banana-Smoothie-%5BRachel-Tsao%5D.png)














































